
"This means that there is a chance that maleate could be degraded by certain substances," says Savchenko. 
Fumarate and succinate, on the other hand, could be more stable, as their HOMO orbitals are equally delocalised. What's more: The analysis also explains why: The electronic density in the HOMO orbital at the C=C bond between carboxylate groups could lead to weaker binding of maleate with molecules or ions. The analysis of the spectral data shows that maleate is potentially less stable than fumarate and succinate. The results can be related to macroscopic properties, especially stability. X-ray absorption spectroscopy (XAS) can be used to investigate the unoccupied electronic states of a system, while resonant inelastic X-ray scattering (RIXS) provides information about the occupied highest orbitals and about interactions between the HOMO-LUMO orbitals. "We analysed these compounds at BESSY II with two different, very powerful methods," says Dr Viktoriia Savchenko, first author of the study. 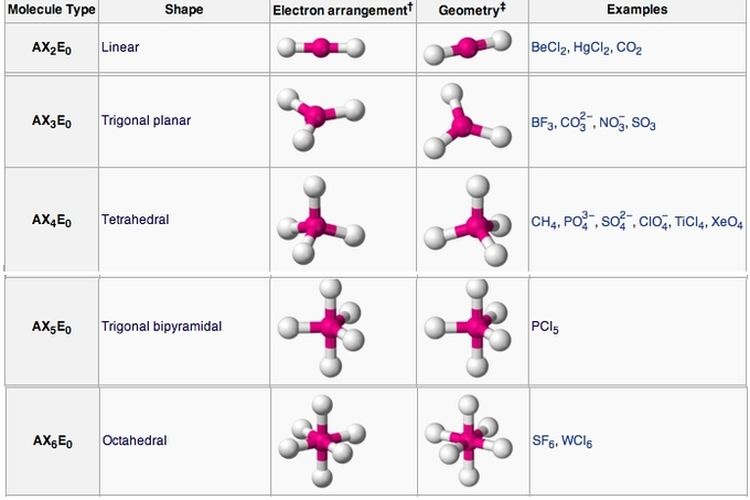
Alexander Föhlisch has elucidated the influence of the electronic structure on the stability of fumarate, maleate and succinate dianions. However, the influence of the molecular orbitals on stability of these molecules has not been researched. The stability of fumarate, maleate and succinate dianions is not only influenced by their molecular geometries, but also by the electronic structure of the molecules, in particular the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). The hybridization of phosphorous in PBr 5 is sp 3 d. The electron geometry of PBr 5 is also Trigonal bipyramidal. In the PBr 5 Lewis dot structure, a total of 15 lone pairs and 5 bond pairs are present. For environmental reasons, however, the question arises as to whether these compounds last forever or are biodegradable. The molecular geometry or shape of PBr 5 is a Trigonal bipyramidal. Check out the article ClF3 Lewis Structure, hybridization, and Geometry. ClF3 is also a similar molecule having its structure as well as hybridization same as IF3. Maleate, on the other hand, which is usually not formed in natural processes, is used in industrial applications that require durable materials. If we check this notation in the VSEPR chart given below we see that IF3 forms a T-shaped molecule. 5 electron groups, 5 bonding groups, 0 lone pairs, 120 equator, 90 axial. 4 electron groups, 2 bonding groups, 2 lone pairs, <<109.5. Fumarate and succinate, for example, are formed as intermediate products in the mitochondria of cells. 4 electron groups, 3 bonding groups, 1 lone pair, <109.5. Some variants are key in coordination chemistry, incorporating metallic elements into organic compounds, other variants play a role in biological processes. Carboxylic acid dianions of the type C 4H 2O 4 or C 4H 4O 4 (fumarate, maleate and succinate) can have different geometries (cis or trans) and different properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
